The Science
Asthma and surfactant protein A (SP-A)
Surfactant is the fluid that lines our airways and helps reduce surface tension as provides host defense.
Surfactant protein A (SP-A) is naturally produced in the lungs and is one of the first lines of defense when encountering a viral infection, environmental irritants or an allergen such as dust mites.
We have discovered that some asthma patients have dysfunctional SP-A, while another large subset has very low to non-detectable levels due to obesity. Based on our pre-clinical studies, we believe our SP-A peptidomimetics would be an ideal therapy for all types of asthma.
Asthma severity is strongly correlated with prolonged eosinophil viability. Eosinophils are prominent in individuals with a Type 2 (T2) driven asthma phenotype, and accrue in large numbers in the circulation, sputum, and airway mucosa. Eosinophil accumulation and prolonged viability in the airways is strongly correlated with greater asthma severity. Indeed, recent studies have shown that within the group of severe asthmatics, 50% have eosinophils present in their lung tissues. Moreover, treatment strategies targeted at reducing eosinophils have been shown to reduce asthma admission rates and exacerbations.
Our work has demonstrated that SP-A induces apoptosis (cell death) of eosinophils isolated from asthmatics and in pre-clinical models of asthma, SP-A therapy led to a significant reduction in lung eosinophilia and while promoting a return to lung homeostasis.
Asthma exacerbations due to infection. One infectious agent, Mycoplasma pneumoniae (Mp), also known as the causative agent of community-acquired pneumonia or “walking pneumonia” has important links to asthma (PMID: 11295645, PMID: 9731038, PMID: 16675358, and PMID: 27401621). Studies have shown that infections with Mycoplasma can precede the onset of asthma, while infections in established asthmatics often result in exacerbations and difficulties with asthma management. While macrolides are recommended for Mp treatment, it is well noted that these are only bacteriostatic against Mycoplasma infection, leaving susceptible asthmatics more likely to experience severe exacerbations. Studies have shown that up to 50% of acute asthma exacerbations were triggered by Mycoplasma and that Mycoplasma may be present in as many as 45% of adult patients with chronic stable asthma. Therefore, a substantial need exists for the development of better therapeutics to target the underlying Mp infection, as well as control of the associated inflammatory phenotypes.
Our work has demonstrated that SP-A-derived small peptides can reduce a major cytokine associated with Mp infection and asthma exacerbations, TNF-a, by inhibition of a major signaling pathway in the nucleus.
SP-A derived small peptide therapeutic
We have identified a small Amino Acid sequence (less than 20 AA) that can recapitulate the function of the full length SP-A in asthma and infection models. We are working to optimize stability and effectiveness through our next-generation SP-A-derived peptidomimetics that are in development. We propose a replacement therapy that utilizes stable and deliverable derivatives of this novel endogenous lung protein.
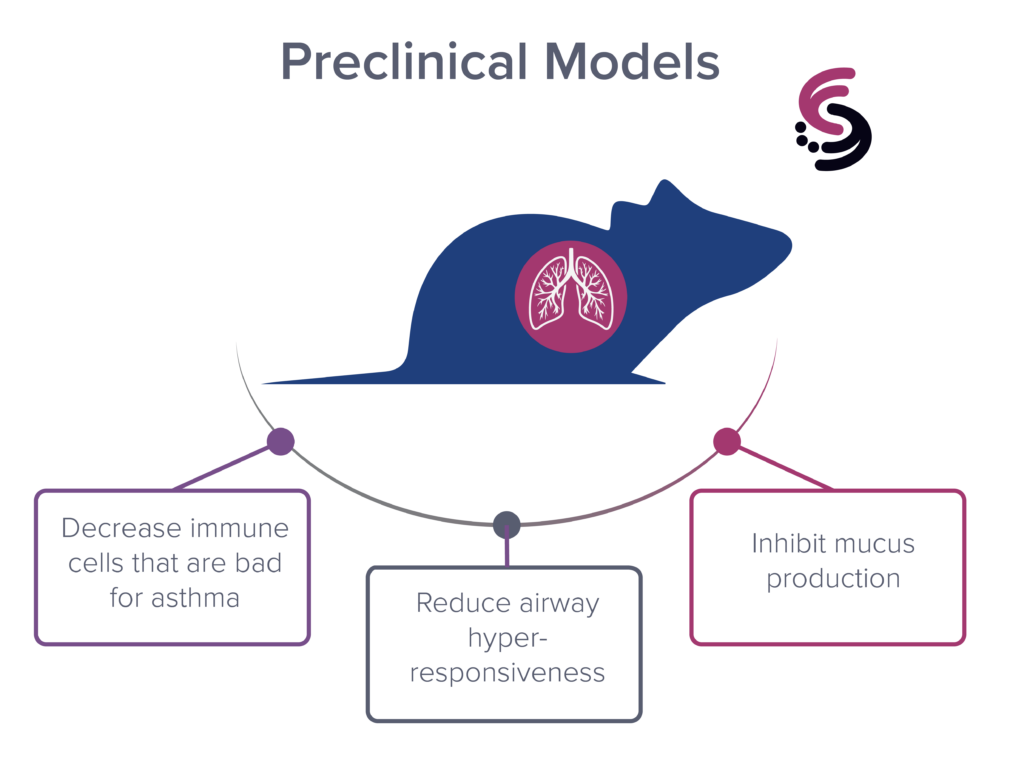
Pre-Clinical Models
In preclinical mouse models, our SP-A peptide has been shown to:
- decrease immune cells that are bad for asthma (eosinophils and neutrophils),
- inhibit mucus production, and
- reduce airway hyperresponsiveness
In human airway epithelial cells, particularly from human cells derived from patients with asthma, as compared to cells from non-asthmatic patients our SP-A peptide has been shown to:
- decrease immune mediators/cytokines,
- decrease mucus production, and
- decrease key cell-signaling pathways upregulated in asthma.
NIH Funding
NIH 2R42HL152942-02 (phase 2)
“Development of SP-A Derived Peptidomimetics for the Treatment of Asthma – Phase II” (Ledford J, Kraft M, MPI)
The goals of this project are to build on the Phase 1 results where we have our identified lead peptidomimetic. We will develop aerosol delivery and perform toxicology studies to submit an Investigational New Drug application to the FDA and present a new class of therapeutics for asthma and other inflammatory lung diseases.
NIH HLR41HL152942-01 (STTR phase 1)
“Development of Surfactant Protein A Peptidomimetics for the Treatment of Asthma” (Kraft M, Ledford J, MPI)
Funded to RaeSedo, LLC with subcontract to University of Arizona
7/1/2020-6/30/22
The goal of this project is to screen and identify lead peptidomimetics of surfactant protein A for treatment of asthma and other inflammatory lung diseases.
NIH AIU19AI125357 Asthma and Allergic Diseases Research Center
“Dysfunction of Innate Immunity in Asthma (Kraft M, Overall PI; Ledford J, co-investigator);
renewed and now titled: “Viral Infection and Innate Immunity in Asthma”
9/1/2015-8/30/2026
The goal of this program is to evaluate the roles of surfactant protein A, tollip interacting protein and palmitoyloleoyl phosphatidylglycerol (POPG) as innate immune modulators in asthma, particularly in the setting of viral infection.
NIH R21 AI135935
“Surfactant Protein A as a Novel Therapeutic for Asthma” (MPIs: Ledford J, Mansour H; Kraft M, co-investigator)
12/05/2017-11/30/2019
Our novel hypothesis is that a specific 10 AA sequence within the carbohydrate recognition domain (CRD) of SP-A confers the ability to significantly suppresses allergic inflammation.
Role: Co-Investigator
Arizona Biomedical Research Grant (ADHS16-162519).
“Surfactant Protein A as an Innate Immune Modulator in Asthma” (Kraft M, PI; Ledford J, co-investigator)
7/1/2017-6/30/2020
The goal of this proposal is to verify the hypothesis that in asthma, dysfunction of SP-A, due to quantitative and functional deficiencies in the protein, is associated with reduced ability to modulate inflammation. This dysfunction results in increased allergic inflammation in asthma. We propose that the basis of dysfunction is both genetic and structural.
NHLBI 1R01 HL125602
“Surfactant Protein A Regulation of Eosinophil Apoptosis in Asthma.” (Ledford J, PI, Kraft M, co-investigator)
7/1/2014-6/30/19
The goal of this proposal is to gain an understanding of how SP-A eosinophil induces apoptosis and allergic airways resolution.
NIH P01-AI081672
“Surfactant Protein A Regulation Immunity in Asthma” (Kraft M, Overall PI, Ledford J, co-investigator)
7/1/2009-6/30/2014
The central hypothesis tested was that SP-A which normally regulates innate immunity and protects the host from persistence and exacerbation of asthma, is dysfunctional in asthma.
Patents
RaeSedo Inc. has licensed all intellectual property (IP) from the University of Arizona. We currently have one patent issued (US Patent Application No.: 16/093,063; patent number 11,110,152) as of September 7, 2021, entitled “COMPOSITIONS AND METHODS FOR TREATING AND PREVENTING LUNG DISEASE.” The Inventors are Dr. Julie Ledford (PhD; Co-Founder and CSO of RaeSedo Inc.) and Dr. Monica Kraft (MD; Co-Founder and CMO of RaeSedo Inc.). On this first patent, we have also filed a continuation in progress (CIP)-Patent Cooperation Treaty (PCT) on August 23, 2022, Inventors are Drs. Ledford, Kraft and Vagner.
We have an additional patent pending (International Patent Application No.: PCT/US2021/026411) which was filed April 8, 2021, that claims priority to 63/006,831 “COMPOSITIONS AND METHODS FOR TREATING AND PREVENTING LUNG DISEASE” and is entitled: “SP-A PEPTIDE MIMETICS IN THE TREATMENT OF INFLAMMATORY LUNG DISEASES.” Inventors on this latter application include Julie Ledford, Ph.D.; Monica Kraft, M.D.; and Josef Vagner, Ph.D., Scott Boitano Ph.D. This application contains the peptidomimetics we intend to take to market. We will bring National Stage filings in Oct 2022.
Publications
1) Francisco D, Wang Y, Conway M, Hurbon AN, Dy ABC, Addison KJ, Chu HW, Voelker DR, Ledford JG, Kraft M. Surfactant Protein-A Protects against IL-13-Induced Inflammation in Asthma. J Immunol. 2020 May 15;204(10):2829-2839. doi: 10.4049/jimmunol.1901227. Epub 2020 Apr 3. PMID: 32245819; PMCID: PMC7304346
2) Younis US, Chu HW, Kraft M, Ledford JG. A 20-Mer Peptide Derived from the Lectin Domain of SP-A2 Decreases Tumor Necrosis Factor Alpha Production during Mycoplasma pneumoniae Infection. Infect Immun. 2020 Aug 19;88(9):e00099-20. doi: 10.1128/IAI.00099-20. Print 2020 Aug 19.PMID: 32513852, PMCID: PMC7440763
3) Dy, A.B.C, Langlais, P.R., Barker, N.K., Addison, K.J., Tanyaratsrisakul, S., Boitano, S., Christenson, S.A., Kraft, M., Meyers, D., Bleecker, E.R, Li, X., Ledford, J.G. “Myeloidassociated differentiation marker is a novel SP-A-associated transmembrane protein whose expression on airway epithelial cells correlates with asthma severity.” Scientific Reports. 2021 Dec 3:11(1):23392. PMC2188087
4) Pederson WP, Cyphert-Daly JM, Tighe RM, Que LG, Ledford JG. Genetic variation in surfactant protein-A2 alters responses to ozone. PLoS One. 2021 Feb 22;16(2):e0247504. doi: 10.1371/journal.pone.0247504. eCollection, 2021. PMID: 33617569, PMCID: PMC7899376
5) Francisco, D., Wang, Y., Marshall, C., Conway, M., Addison, K.J., Billheimer, D., Kumura, H., Numata, M., Chu, H.W., Voelker, D.R., 1Kraft, M, 1Ledford, J.G. Small peptide derivatives within the carbohydrate recognition domain of SP-A2 modulate asthma outcomes in mouse models and human cells. 1Co-senior authors. Frontiers Immunology, epub ahead of print, July 8, 2022.
